KUBio BSL-2 生产解决方案
通过模块化解决方案加快疫苗制备工作
无需等待,立即使用 KUBio BSL-2 选项制作病毒载体
随着病毒载体产能缺口不断增加,使得生产商不得不评估并比较购买模式与自建模式下的关键生产策略。我们可以为您提供快速灵活,全面的解决方案。根据您的产能需求,选择 KUBio BSL-2 模块化设施或 KUBio 箱为病毒载体。两种端到端解决方案均可放大,包括 FlexFactory 一次性工艺线、集成的 Figurate 智能化平台以及经过验证的工艺支持和专业知识。
了解更多
加速基于病毒载体疗法的生产
我们可在短短 10 个月内为您的研究中心提供 KUBio 病毒载体溶液盒。该交钥匙式包装设计用于有效生产针对腺病毒和慢病毒的生物疗法药物。此生物安全二级 (BSL-2) 的环境包括专有的 FlexFactory 病毒载体一次性工作流程,旨在实现灵活操作并降低污染风险。您可以放大 KUBIO 盒的占地面积以满足生产需求的变化。
了解更多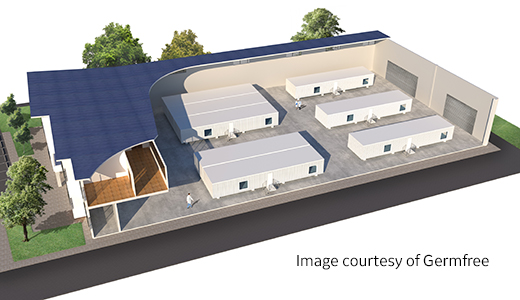
在安全环境中推进大规模生产
当您的生产计划有大规模生产需求时,the KUBio™ BSL-2 模块化设施可仅用 18 个月以内的时间提供灵活的可扩展解决方案。 实现灵活性并凭借速度和效率赢得自信。KUBio™ BSL-2 设施包括 FlexFactory™ 一次性生物制造平台, Figurate™ 集成智能化和工艺支持, 您可以搭建第二个完整的工艺环境,把您的占地面积从 2350 m2 扩大至 3850 m2。

灵活性让您能够确保进度和缓解风险
产能扩增决策通常需要在风险较高的药物生命周期早期进行投资。KUBio 模块化设施通过并行模块、工艺设备和现场工作将设施搭建时间压缩了 50%。您可以推迟资本支出或投资到其他地方,直至您获得更高的成功机会。
浏览 KUBIO 解决方案的益处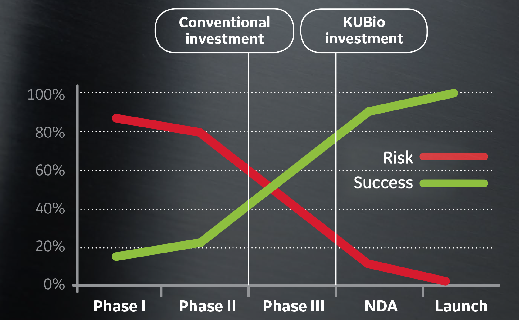
一次性技术的灵活性
病毒载体和病毒生产需要一次性技术的支持,以实现多产品生产,提高效率,并降低污染风险。用于在鸡蛋中生产疫苗的老技术已经成为历史。凭借集成的 FlexFactory 生产线和 Cytiva 的经验以及相关支持来开启未来。
了解有关 FLEXFACTORY 的更多信息