Chronicle™ automation software connects Cytiva and third-party instruments. From process development through commercial GMP manufacturing, pre-GMP and GMP plans have been designed to support you.
Digitize records to reinforce GMP compliance and improve sample security with Chronicle™ automation software. eSOPs manage deviations, promote adherence, and provide training. Chronicle™ automation software has been independently audited against GAMP5, 21 CFR Part 11 and EU Annex 11. Plus, Chronicle™ automation software can support two different environments—the cell therapy and the biologics environment—to better fit your specific needs.
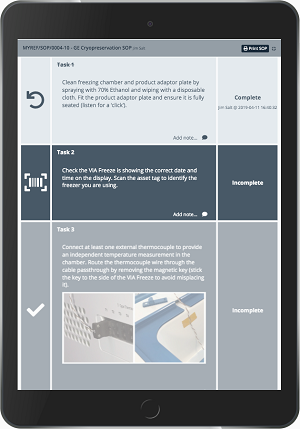
- Implement electronic batch records that comply with regulatory requirements, reviewing deviations efficiently with automated workflows to support release by exception.
- Optimize operators’ time and help reduce mistakes with electronic standard operating procedures (eSOPs).
- Schedule procedures by reserving instruments and checking availability on a built-in calendar.
- Trace all your products and consumables with stock control, barcode label printing, and scanning. Obtain a Certificate of Analysis (COA) automatically for many Cytiva cell therapy reagents and plastics.
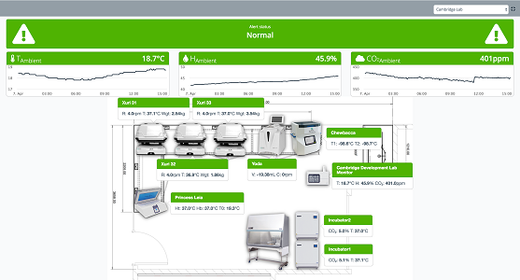
- Connect third-party instruments to upload and visualize instruments data.
Chronicle™ automation software provides a unified digital platform to monitor your facility manufacturing operations and supply chain logistics. Plus, electronic batch records will trace every manufacturing step.
Read more| Product | Product Name | Price | ||
|---|---|---|---|---|
| MYC_60001 |
Chronicle™ automation software GMP hosting setup
|
One-time setup fee | On Request |
|
| 29734893 |
Chronicle™ automation software pre-GMP annual subscription for “unlimited” number of users
|
Annual | On Request |
|
| 29734896 |
Chronicle™ automation software GMP annual subscription for 25 additional users
|
Annual | On Request |
|
| 29734890 |
Chronicle™ automation software pre-GMP base annual subscription
|
Annual | On Request |
|
| 29734895 |
Chronicle™ automation software GMP annual subscription for 10 additional users
|
Annual | On Request |
|
| 29734892 |
Chronicle™ automation software pre-GMP annual subscription for 10 additional users
|
Annual | On Request |
|
| 29734894 |
Chronicle™ automation software GMP base annual subscription
|
Annual | On Request |
|
| 29734897 |
Chronicle™ automation software GMP annual subscription for “unlimited” number of users
|
Annual | On Request |
|
| 29734891 |
Chronicle™ automation software pre-GMP annual subscription for 5 additional users
|
Annual | On Request |
|