
Scaling up bioprocesses through a CDMO
Roivant Sciences and Cytiva’s Fast Trak services collaborate to accelerate an orphan drug to clinic.
Successful chemistry, manufacturing, and control activities streamline drug development without compromising quality or efficacy
Manufacturing novel biotherapeutics poses complex challenges that can make it difficult to meet milestones without taking on unexpected risk. You need a robust process, a secure supply chain, and a way to minimize production deviations so patients receive high-quality therapies each time. Early design choices can affect your overall speed to market. Build your expertise with ours.
Process development Introduction
Manage raw materials | Scale-up | Process development resources | Application solutions
It is not straightforward to scale molecules to meet regulatory and patient demand. While you’ve mastered the science of therapeutic innovation, we’ve mastered the art of bioprocessing, with decades of experience taking companies from start-up to clinicals trial and commercial success. Together, we can help you reduce risk and make good decisions faster.

Roivant Sciences and Cytiva’s Fast Trak services collaborate to accelerate an orphan drug to clinic.
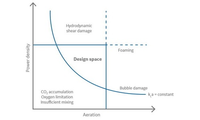
Translating monoclonal antibody production from microscale to 1000 L.

Transferring a process from a laboratory-scale idea to GMP-compliant commercial product.
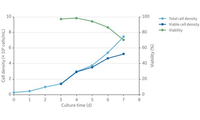
Simple batch process scales in linear manner from 5 L to 28 L in single-use bioreactors.

Process intensification and other approaches to increase efficiency.
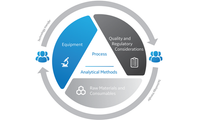
Don’t risk reducing the quality and efficiency of your process during handoff.
Learn more about key considerations and strategies in mRNA manufacturing workflow.

Upstream, downstream, and analytics support to take you from idea to pure characterized product.

Technologies to close, automate, and digitize your cell therapy process for consistent and scalable production.

Flexible, scalable solutions to help you quickly add capacity to develop and manufacture your own plasmids and viral vectors.
Manufacturing approaches for novel vaccines to include vaccines that are bacterial, virus, protein polysaccharide or DNA-based.

Balancing innovation, speed, and robustness to support biotherapeutic production.