Why does your lab filtration suddenly slow down or stop?
Have you ever been in the middle of an important experiment when your filtration process grinds to a halt? It’s frustrating, time-consuming, and can throw off your entire workflow. This common issue known as laboratory filter blocking, can significantly impact the efficiency and accuracy of your lab work. What exactly causes it, and more importantly, how can you prevent it? Could your filter be the culprit?
When troubleshooting laboratory filter blocking, we often look at the sample or the setup. But have you ever considered how the structure and material of your filter might be contributing to the problem?
Understanding surface membrane filters
Surface membrane filters are the workhorse of many laboratory filtration systems. These filters are typically thin and made from highly porous materials with a narrow pore size distribution. Their job is to retain particles larger than their rated pore size and prevent them from passing through. In life science applications, the most used membrane materials are:
- Polyethersulfone (PES)
- Polyvinylidene fluoride (PVDF)
- Nylon
- Cellulose acetate
- Regenerated cellulose
Each of these materials has its own unique properties. Nylon is highly durable in harsh chemical environments but tends to bind proteins—something you want to avoid in protein-sensitive applications. PES and PVDF are prized for their low protein binding and are suited for protein purification and analysis, but may degrade in the presence of organic solvents.
How are these filters made?
These membranes are typically manufactured using solvent casting or immersion casting techniques. In both methods, a polymer solution is exposed to a non-solvent, causing the polymer to precipitate and form a solid membrane, as shown by closeup view of PES in Figure 1. This process allows for precise control over pore size and distribution.
Fig 1. Microscopy image of PES
By tweaking the casting conditions, manufacturers can produce membranes with a range of pore sizes—most commonly between 0.8 µm and 0.1 µm—to suit different filtration needs. A summary of these options is shown in Table 1.
Table 1. List of membrane pore sizes typically found in microfiltration membranes
| Pore Size | Level of filtration |
| 0.8 | Pre- filtration |
| 0.45 | Clarification |
| 0.2 | Sterile filtration |
| 0.1 | Sterile filtration and mycoplasma reduction |
Pore size
The top priority in membrane filter design is minimized and controlled pore size variation for consistent and reliable retention at the specified rating. A wide distribution of pore sizes can compromise the filter’s claimed retention performance. To validate that a 0.2 µm filter qualifies as sterilizing-grade, it must completely retain Brevundimonas diminuta when challenged with a concentration of 10⁷ cfu/cm² of effective filtration area. Only filters that achieve 100% retention under these conditions can be classified as sterilizing-grade. Due to these stringent standards, membrane capacity becomes a secondary consideration. Fortunately, other filtration media, such as depth filters, are better suited for handling high particulate loads upstream of sterile filtration. These prefilters protect the final membrane and optimize overall filtration performance.
During filtration, membrane blockage can occur with particles both larger and smaller than the pore size. Over time these particles will accumulate on the surface or within the pores of the membrane, which significantly reduces flux and eventually completely stops the flow of filtrate. The various ways this occurs can be explained in more detail by the laws of blocking filtration (1).
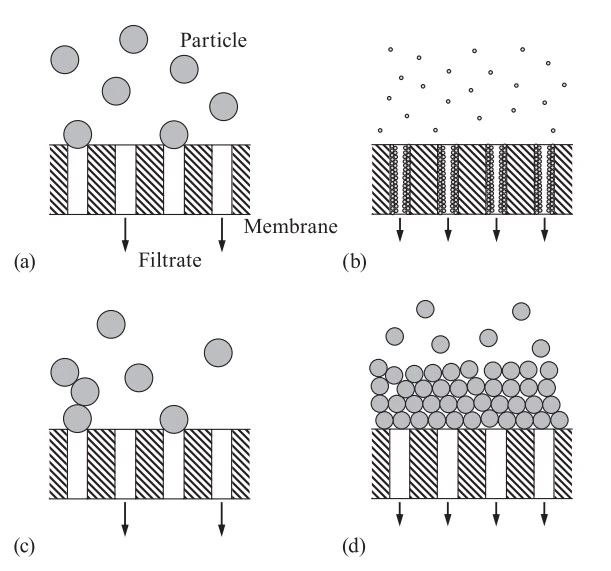
Blocking filtration laws are applied to four different fouling patterns for describing the deposit of particles on filter media and membranes (Fig 2).
- Complete block: Large particles block pores at the surface. Once a pore is blocked, no liquid can pass through it.
- Intermediate blocking: Similar to complete blocking, but not every particle blocks a pore. Some pass through, while others clog the surface.
- Standard blocking: Smaller particles stick to the inside walls of the pores. This slowly narrows the pores and reduces flow over time.
- Cake formation: Instead of blocking the pores, particles pile up on the surface of the filter forming a thick layer or “cake.” As this cake grows, it becomes harder for liquid to pass through.
Solutions that contain large amounts of particulate, such as cell culture solutions, can quickly block a surface filter and reduce filtrate flow resulting in filter replacement earlier than desired.
Fig 2. Schematic view of four fouling patterns in blocking filtration laws: (a) complete blocking law, (b) standard blocking law, (c) intermediate blocking law, and (d) cake filtration law
To overcome this undesired effect, the need for sterilization must be balanced with maintained flowrate and throughput. This can be done by prefiltering high particulate samples.
Solutions derived from cell cultures, fermentation broths, and cell lysates often contain large amounts of particulate load or debris. The clarification of these solutions with a prefilter removes large particles prior to further downstream processing. This protects the surface membrane filter for micro- and ultrafiltration or capture chromatography.
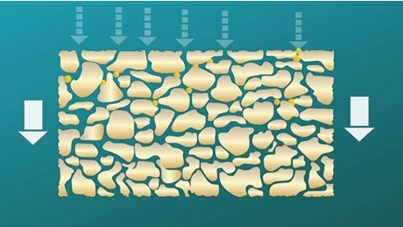
Clarification, also known as prefiltration, is performed with devices containing depth filter media such as glass fiber, cellulose fiber, and polypropylene. This media is more efficient because a third dimension, thickness, is added to the filter to trap particulate matter both at the surface and through the inside of the entire filter media. In Figure 3, a depth media schematic is compared to a membrane media schematic.
(a)
Fig 3. Schematic of (a) depth media and (b) membrane media.
(b)
Depth media is manufactured following a wet-laid nonwoven paper making process that creates a matrix of randomly distributed fibers. This results in tortuous paths with pores of undefined size and shape. The matrix fibers that provide structure to wet-laid media can be cellulosic, glass or polyolefine fibers. To enhance particle filtration and adsorption, certain additives can be incorporated, such as perlites, diatomaceous earth, and polyvinylpolypyrrolidone (PVPP). This complex matrix can retain particles using two mechanisms: Adsorption and size exclusion. Different depth media substrates are layered to effectively capture debris and particulate matter. This gives depth filtration devices a much higher hold capacity for debris capture than is possible with a surface membrane and more solution can be filtered.
This high holding capacity can be seen in the our AcroPrep™ 24-well filter plate that clarifies and sterilizes within a single device and workflow step. Using either a vacuum manifold or centrifuge, high density cell cultures such as CHO or HEK can quickly be processed to capture of cells, cell debris, and other biological aggregates in the filter media. The filtrate collected contains proteins and other sub 0.2 μm particles. This innovative workflow is possible with multi-layer filtration media and membranes integrated into one device.
The top layer features Pall’s Seitz depth media and efficiently captures whole cells and cell debris. The lower Supor™ EKV membrane layer provides an efficient sterile filtration layer. Supor EKV membrane is made of hydrophilic polyethersulfone that is low in protein binding and offers excellent throughput due to its asymmetric structure.
Understanding the mechanisms behind laboratory filter blocking and the differences between surface membrane and depth media filtration is essential to optimize laboratory workflows. By incorporating effective prefiltration strategies—especially when working with high-particulate solutions like cell cultures and fermentation broths—researchers can significantly extend membrane lifespan, maintain consistent flow rates, and gain reliable results. Thoughtful filter selection and system design improves efficiency and safeguards the integrity of critical experiments. While not all filter blocking is inherently negative—it can indicate effective particle retention—efficiently removing particulates and extending the life of membrane filters is essential to sustained performance. Visit our website and learn more about our depth and membrane filter options to find Cytiva solutions tailored to you!
REFERENCES
- Iritani E, Katagiri N. Developments of Blocking Filtration Model in Membrane Filtration. KONA Powder and Particle Journal. 2016;33(0):179-202. doi: 10.14356/kona.2016024
Cytiva and the Drop logo are trademarks of Life Sciences IP Holdings Corporation or an affiliate doing business as Cytiva.
AcroPrep and Supor are trademarks of Global Life Sciences Solutions USA LLC or an affiliate doing business as Cytiva.
© 2025 Cytiva