Standardize vs customize: the single-use consumable conundrum
Single-use bioprocessing technology offers clear advantages: it allows for greater flexibility in facility design, helps reduce contamination risks, reduces time between batches, and can be more affordable upfront than some reusable systems. However, recent surges in demand have exposed a key vulnerability—its reliance on a stable supply chain. Because single-use systems depend on a continuous flow of consumables, any disruption to the supply chain can lead to process and delivery delays. Unlike large-scale reusable equipment, which is typically a one-time investment, single-use systems also require ongoing engagement with suppliers throughout the equipment’s lifecycle.
These two factors—potential for supply chain disruptions and the closer relationships between biomanufacturers and suppliers—are closely linked. How your single-use supplier manages their inventory and responds to disruptions directly affects delivery timelines to your facility.
Beyond logistics, the quality and performance of the consumables themselves (ease of use, process performance, and durability) play a critical role in the success of a process. Operator error, for example, is a common cause of batch failure (Fig 1). Personnel training is one part of the solution for this, but suppliers can also help by designing equipment that is easier to use.

Fig 1. Estimated average rates of batch failure by cause in 2024 as reported to BioPlan Associates (1). For consumables such as bioreactor bags, ease of use and reliability can help minimize operator errors, equipment failure, and other process challenges.
There’s a tension here between the need for speed and the requirement for quality. Standardized single-use consumables can help avoid supply-chain delays by being readily stocked and made available on demand. Yet many biomanufacturers still opt for custom designs, even though doing so can extend timelines—both during the initial design and specification phase and later during delivery. The reason? Customization allows the biomanufacturer control over material designs and specifications, whereas standardization shifts that burden to the supplier.
So, is customization still the best approach? (Spoiler alert: we don’t think so – at least not for bioreactor bag assemblies.) We’ll explore recent developments in cell culture technologies and how they’ve made standardization of bioreactor bags more feasible at every scale. We’ll show some examples of how we developed our standardized bag designs, based on more than 20 years of experience, for our newest bioreactor platform, and describe what biomanufacturers should look for when choosing a single-use supplier.
A brief history of bioreactor bag supply models
Within biopharma process development, small, multi-use bioreactor systems (up to 100 L) have been available as commercial-off-the-shelf the shelf (COTS) products. Suppliers have historically achieved this using build-to-stock (BTS) or configure-to-order (CTO) production models. These models enable ready availability of a variety of technologically and performance-capable products, which allow buyers to select, receive, and implement them within development timelines. In effect, this gives buyers the ability to choose the best combination of technical capabilities and process performance.
Historically, the COTS model has not translated to larger-scale pilot or full production volumes systems. At these larger sizes (e.g., >500 L bioreactor volumes), an engineered-to-order (ETO) model is more common, as it helps to address the complex requirements associated with biotherapeutic production capacity and regulatory compliance. This largely perpetuates the single-product, dedicated facility production paradigm.
So what has changed?
Today, engineered-to-order systems are no longer the only viable option for large-scale upstream bioprocessing. Advances in technology and higher process titers have reduced the need for large terminal bioreactor volumes, making single-use bioreactors a practical alternative to biotherapeutic-specific production facilities and the ETO-based equipment.
These revolutionary forces not only support the conventional production models, but they also better enable sequential and simultaneous multi-product production models. The widespread adoption of single-use bioreactors within the biopharmaceutical industry—85% of biomanufacturing facilities use single-use bioreactors in every stage of manufacturing (2)—highlights their value. Consequently, standardization is more practical, from process development to production scales. More importantly, standardization enables COTS (or COTS-like) sourcing of scalable bioreactors platforms all the way into production scale—with potential gains in speed and agility for manufacturers.
The foundations of standardization
For suppliers, standardization can simplify inventory management and offer greater control over delivery timelines to customers. To standardize effectively, suppliers ultimately need to collate and condense data from a variety of sources into a coherent and actionable design plan that will suit a variety of use cases and applications (Fig 2). For this, they need extensive experience in similar or identical products, a robust quality management system (QMS), and sufficient manufacturing expertise.
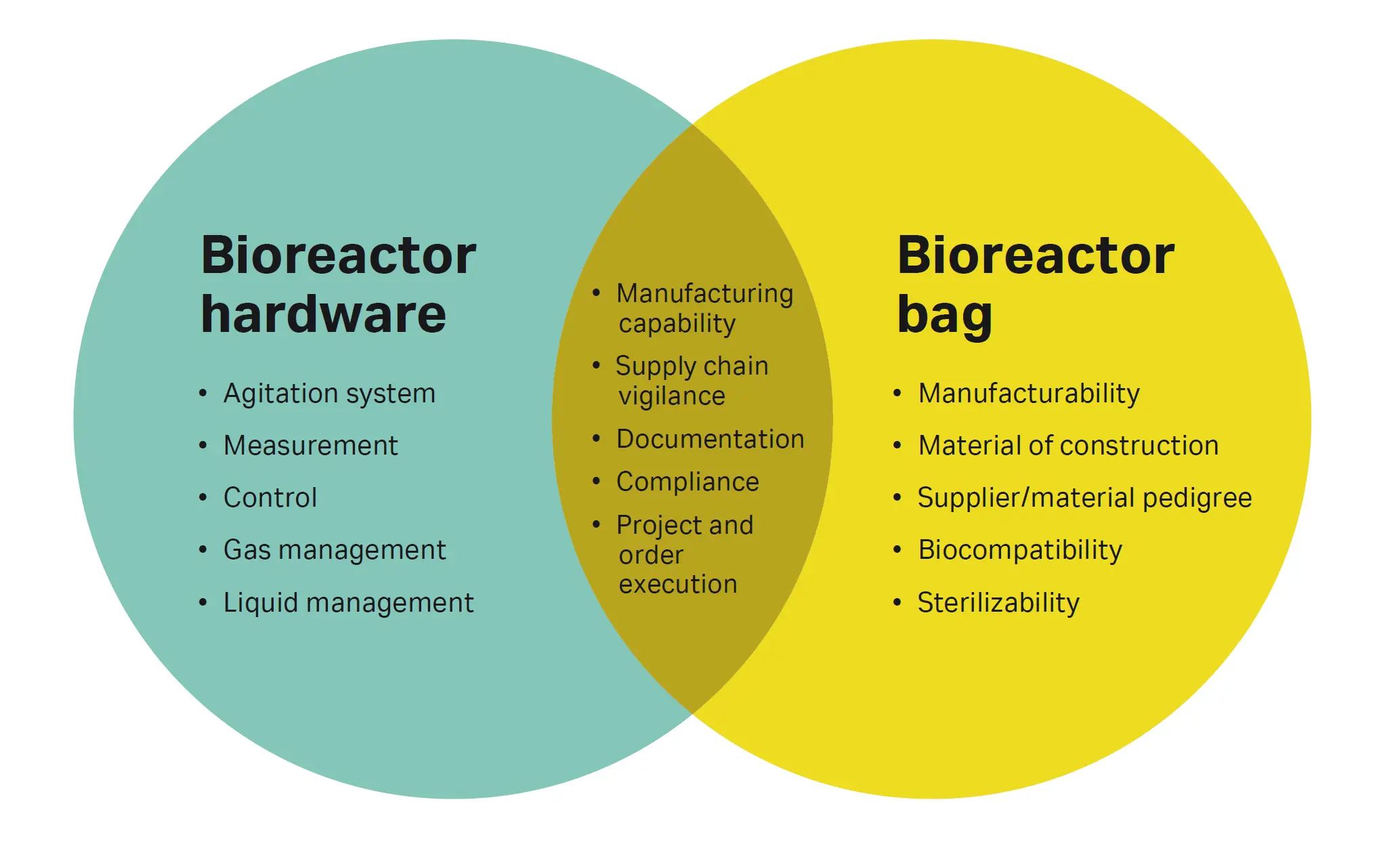
Fig 2. Single-use bioreactor interdependent design spaces. How the design space is defined will determine if a standard, configurable or customized design is required.
Supplier QMS provides the foundation for effective supply chain management, design, and change controls, complaint handling, and overall product quality. Manufacturing expertise comes from the refinement of procedures and methods, leveraging the economies of scale in purchasing, inventory, and logistics. This results in more reliable products and dependable deliveries.
Customer feedback spanning a broad spectrum of applications and use-cases, and the technical ability to transform that feedback into meaningful data, is also necessary for effective standardization. All these insights can be used to inform the design of a product platform that suits a wide variety of applications and needs (Fig 3).
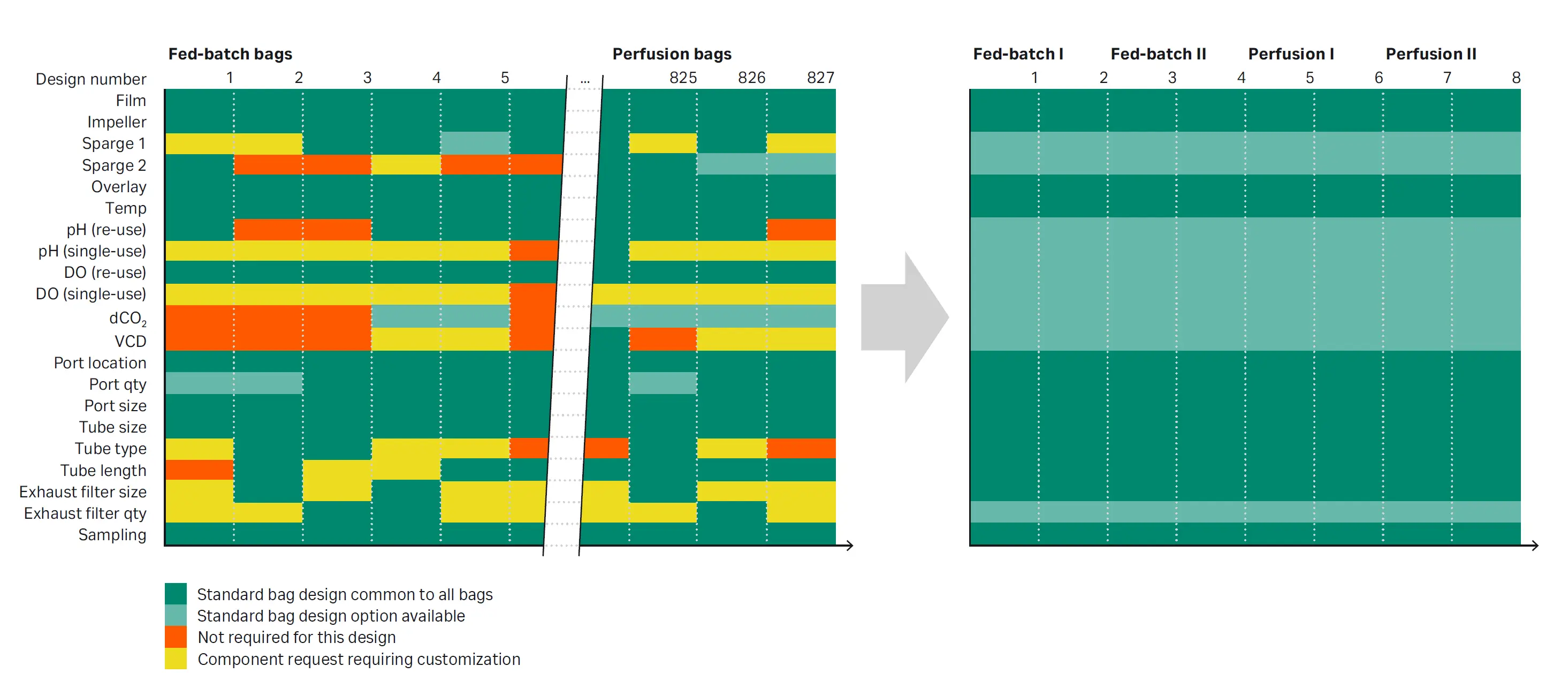
Fig 3. Representation of bag design consolidation. We consolidated around single components where multiple variants of the same thing were present. Standardizing on tube lengths, exhaust gas filter sizes and performance requirements provides the benefits of custom with the simplicity of standard.
Standardization of Xcellerex™ X-platform bioreactors, for example, emerged from over 20 years of experience with the predecessor system, the Xcellerex™ XDR single-use bioreactor. That experience spans thousands of bioreactors, used in process development and biomanufacturing, and several hundred customized single-use bioreactor bags, deployed on automation platforms made by Rockwell, Emerson, and Siemens.
This experience with a vast variety of bioprocess applications, and continual, current customer feedback allowed the development team to consider every aspect of the single-use bioreactor bag, from component selection and location on the bag, to inter- and intra-process connectivity and bag integrity, to supply chain and manufacturing robustness, and inventory management and logistics.
All this enabled us to distil the entire catalog of standard and customized XDR single-use bioreactor bag configurations to define a set of application-centric configurations. The result was a set of standardized bioreactor bags based on three feeding strategies: batch, fed-batch, and perfusion (Fig 4). Along with the bioreactor vessel and software, these designs are intended to maximize operational efficiency and deliver predictable, scalable performance.
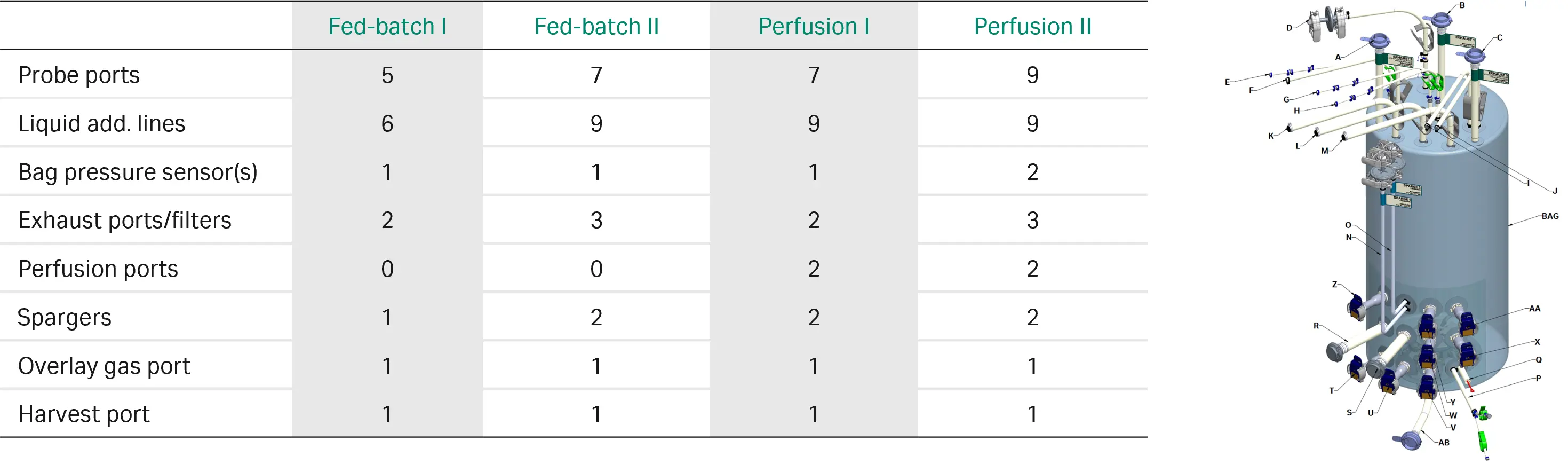
Fig 4. Xcellerex™ X-platform standard bioreactor bag configurations.
As the biopharmaceutical industry continues to advance, the need for speed, reliability, and scalability has never been greater. Standardized bioreactor bags offer a compelling solution that balances operational efficiency with supply chain resilience. And standardization doesn’t mean compromising performance. It means building smarter, more resilient systems in collaboration with experienced suppliers. With the right platform and the right vendor, biomanufacturers can streamline operations, reduce variability, and maintain consistent product quality.
References
- BioPlan Associates. 21st Annual Report and Survey of Biopharmaceutical Manufacturing Capacity and Production: a Study of Biotherapeutic Developers and Contract Manufacturing Organizations. BioPlan Associates, Inc.; 2024.
- Denni I. Report: Single-use bioreactor uptake plateaued, other SUT on the rise. Bioprocess Online. August 12, 2024. https://www.bioprocessonline.com/doc/report-single-use-bioreactor-uptake-plateaued-other-sut-on-the-rise-0001. Accessed May 1, 2025.