常见问题
See the table below for differences among the products:
| Products | HisTrap HP | HisTrap FF | HisTrap FF crude | HisTrap excel |
|---|---|---|---|---|
| Product codes | 17524701,17524705, 17524801,17524802, 17524805,29051021 |
17531901, 17531902, 17525501, 17525502 | 11000458, 11000459, 17528601, 17528602, 29048631 | 17371205, 17371206, 29048586 |
| Base matrix | Ni Sepharose High Performance | Ni Sepharose 6 Fast Flow | Ni Sepharose 6 Fast Flow | Ni Sepharose excel |
| Average bead size | 34 µm | 90 µm | 90 µm | 90 µm |
| Binding capacity (mg /ml) | > 40 mg | Approx. 40 mg | Approx. 40 mg | > 10 mg |
| Application | The high protein binding capacity and the small bead size ensures high-resolution and minimal sample dilution. | The flow properties of the highly cross-linked agarose matrix make it an excellent choice for purification scale-up. | Application of unclarified sample is possible; no need to filter the sample before loading. | Purification of histidine-tagged proteins from samples that can cause Ni stripping from the medium, e.g. secreted proteins in liquids containing chelators. |
Accessories
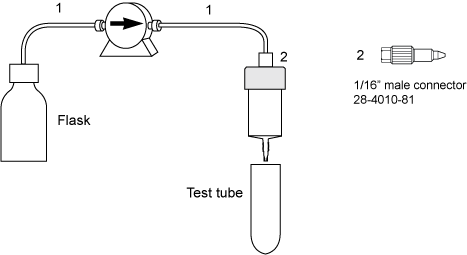
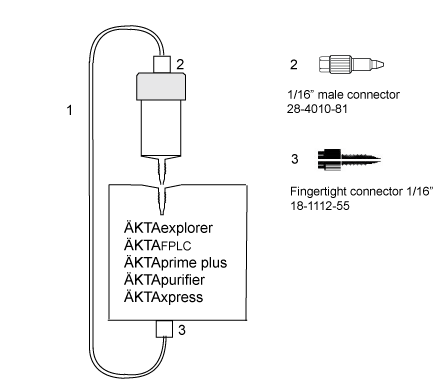
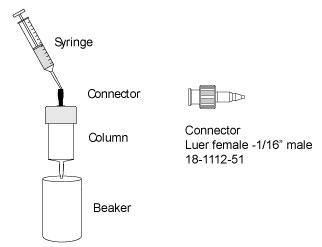
故障排查
寻找产品相关问题的解决方案。对于未列出的问题,请联系当地的Cytiva服务代表。
Back pressure increases during operation
| 可能原因 | 建议的补救措施。 |
|---|---|
Auxiliary equipment such as manometers and pumps not working properly. |
Check the function of all auxiliary equipment. Repair/replace if necessary. |
Sample loading is performed at too high flow rate. |
Decrease flow rate during sample loading. |
Sample viscosity is too high when purification is performed at lower temperature than room temperature. |
Move to room temperature if possible to reduce the sample viscosity. |
The prefilter might be blocked. |
Check the prefilter. |
Valve not fully open. |
Check all valves. Open any that is not fully open. |
Bent tubing. |
Check that the flow path is not restricted. |
Buffer viscosity too high. |
Check the viscosity of all buffers. Viscosity is a function of temperature. (Lower temperature gives higher viscosity.) Let low-temperature buffer reach operating temperature before starting the run. |
Cell paste has too high viscosity. Cell paste not diluted enough. |
Increase dilution of cell paste before sonication or dilute after the sonication to reduce the viscosity. |
Increased precipitation and aggregation. |
Freeze/thaw of the unclarified lysate may increase precipitation and aggregation. Sonication of the thawed lysate can prevent increased back pressure problems when loading on the column. |
Lysate too viscous due high concentration of host nucleic acid. |
1. Continue sonication until the viscosity is reduced and/or add an additional dose of DNAse and Mg2+ According to instructions. |
Mechanical cell disruption is insufficient. |
Increase the efficiency of mechanical cell disruption (for example increase sonication time). Keep the sample on ice to avoid frothing and overheating as this may denature the target protein. |
Microbial growth in buffers. |
Check buffers, especially those with phosphate, for microbial growth. Replace with fresh buffer if necessary. |
Sample and collection vessels at different levels |
Adjust the vessels to approximately the same level. |
Unsatisfactory elution
| 可能原因 | 建议的补救措施。 |
|---|---|
Partial degradation of tagged protein by proteases. |
Add proteases inhibitors. |
Contaminants have high affinity for nickel ions. |
Elute with a stepwise or linear imidazole gradient to determine optimal imidazole concentrations to use for binding and for wash; add imidazole to the sample in the same concentration as the binding buffer. Wash thoroughly before elution with binding buffer containing the highest possible imidazole concentration (chosen imidazole concentration must not cause elution of the histidine-tagged protein). A shallow imidazole gradient (20 column volumes or more), may separate proteins with similar binding strengths. If optimized conditions do not remove contaminations, further purification by ion exchange chromotagraphy (HiTrap Q HP or HiTrap SP HP) and/or gel filtration (Superdex peptide, Superdex 75 or Superdex 200) may be necessary. |
Contaminants are associated with tagged proteins. |
Add detergents and/or reducing agents before sonication cells. Increase detergent levels (e.g. up to 2% Triton X-100 or 2% Tween 20) or add glycerol (up to 50%) to the wash buffer to disrupt nonspecific interactions. |
| 可能原因 | 建议的补救措施。 |
|---|---|
Buffer/sample composition is not optimal. |
Check pH and composition of sample and binding buffer. Ensure that the concentration of chelating or strong reducing agents in the sample as well as the concentration of imidazole is not too high. |
Column capacity is exceeded. |
If HisTrap 1 ml columns have been used, change to larger column, HisTrap 5 ml. |
Histidine tag is partly obstructed. |
Purify under denaturing conditions. Use 4-8 M urea or 4-6 M Gua-HCl. |
| 可能原因 | 建议的补救措施。 |
|---|---|
Dead volume in chromatography systems is high. |
Minimize dead volume in the chromatography system by decreasing capillary length and dimensions between injector and detector. Bypass unused system components e.g. column valves from the flow path. |
Flow velocity too high. |
Run the separation at a lower flow velocity. This is especially important for adsorbents that bind several substances and where selectivity is low. |
Too much sample has been loaded onto the column. |
Decreasing the sample load may improve the resolution significantly. |
| 可能原因 | 建议的补救措施。 |
|---|---|
Histidine-tagged protein still bound due too mild elution conditions. |
Determine the optimal elution conditions by |
Protein has precipitated in the column. Sample quantity or protein concentration are too high. |
Decrease amount of sample or decrease protein concentration by eluting with linear imidazole gradient instead of imidazole steps. Try detergents or change NaCl concentration or elute under denaturing (unfolding) conditions. Use 4-8 M urea or 4-6 M Gua-HCl. |
Nonspecific hydrofobic or other interactions. |
Add a nonionic detergent to the elution buffer (e.g. 0.2% Triton X-100) or increase the NaCl concentration. |
Protein found in the flowthrough. Concentration of imidazole in the sample and/or binding buffer is too high. |
Decrease imidazole concentration. |
Protein found in the flow through. Histidine tag may be insufficiently exposed. |
Perform purification of unfolded protein in 4-8 M urea or 4-6 M Gua-HCl as for inclusion bodies. To minimize dilution of sample, solid urea or Gua-HCl can be added. |
Protein found in the flow through. Buffer/sample composition is not optimal. |
Check pH and composition of sample and binding buffer. Ensure that the concentration of chelating or strong reducing agents in the sample as well as the concentration of imidazole is not too high. |
Target protein may not be histidine-tagged as expected. |
Verify DNA sequence of the gene. Analyze samples taken before and after induction of expression with e.g. anti-His antibodies in Western blotting. |
| 可能原因 | 建议的补救措施。 |
|---|---|
Target protein not stable under the chosen conditions and partly degrades |
Find better ways to stabilize the protein, e.g. shorten the process time. |
The detergent has formed micelles with the protein, thereby increasing its size and changing its elution position. |
Reduce the concentration of detergent to below it´s critical micelle concentration (CMC) value. |
| 可能原因 | 建议的补救措施。 |
|---|---|
Column not properly equilibrated. |
Check the pH and conductivity of the effluent before applying the sample. Continue to equilibrate with start buffer if necessary. |
Components in the sample displace the target molecule before elution starts. |
Reduce the concentration of detergent to below it´s critical micelle concentration (CMC ) value. |
| 可能原因 | 建议的补救措施。 |
|---|---|
Wrong additives may be used. |
Try additives recommended in instructions |
Protein might be insoluble (inclusion bodies) |
Protein can usually be solubilized (and unfolded) from inclusion bodies using common denaturants such as 4-6 M Gua-HCl, 4-8 M urea or strong detergents. Prepare buffers according to instructions |
| 可能原因 | 建议的补救措施。 |
|---|---|
Wrong additives may be used. |
Try additives recommended in instructions |
| 可能原因 | 建议的补救措施。 |
|---|---|
The column not properly equilibrated |
Check the pH and conductivity of the effluent before applying the sample. Continue to equilibrate with start buffer if necessary. |
Column has clogged
| 可能原因 | 建议的补救措施。 |
|---|---|
Sample pretreatment has been insufficient. |
If cleaning-in-place, according to instructions, is unsuccessful, replace the column. Preventive action for HisTrap HP and HisTrap FF: |
Column leakage
| 可能原因 | 建议的补救措施。 |
|---|---|
Connectors not compatible with each other. |
Check compatibility. |
Connectors not compatible with solvents. |
Check chemical resistance with the connector supplier. |
Connectors poorly positioned or not tightened. |
Check the connectors. |
Gaskets worn out. |
Gaskets lose flexibility with time and need to be replaced regularly. Inspect and replace if necessary and at least annually. |
| 可能原因 | 建议的补救措施。 |
|---|---|
Tubing not compatible with solvents. |
Check chemical resistance with the tubing supplier. |
Poor reproducibility
| 可能原因 | 建议的补救措施。 |
|---|---|
Bound substances not removed during cleaning. |
Clean the chromatography medium according to instructions. |
Ligand partially degraded. |
Replace the chromatography medium. |
| 可能原因 | 建议的补救措施。 |
|---|---|
Affinity, hydrophobic interaction and ion exchange chromatography - Continuous build-up of contaminants has altered the selectivity of the chromatography medium. |
Clean the chromatography medium according to instructions. |
| 可能原因 | 建议的补救措施。 |
|---|---|
Column not properly equilibrated |
Check the pH and the conductivity of the effluent before applying the sample. Continue to equilibrate with start buffer if necessary |
Insufficient column regeneration. |
Prolong the regeneration. |
| 可能原因 | 建议的补救措施。 |
|---|---|
Protein properties change with concentrations |
Dilute or concentrate the sample to minimize effects. |
Proteins precipitate at high concentration. |
Reduce sample concentration and/or binding capacity. |
| 可能原因 | 建议的补救措施。 |
|---|---|
Column bleeding from previous run. |
Check and adjust your cleaning procedure. |
Column clogged with denatured proteins and/or lipids. |
Clean and regenerate the column and chromatography medium according to instructions. |
Incomplete equilibration of the column. |
Check pH and conductivity of the effluent before applying the sample. Continue to equilibrate if necessary. |
Incorrect pH and/or ionic strength of the solutions. |
Check pH and conductivity and adjust if necessary. Calibrate your conductivity and pH meters. |
Larger sample mass load applied compared with earlier runs. |
Keep mass of sample constant when repeating runs. (High proteins concentration can cause protein interaction, resulting in change of elution profile.) |
Sample volume is different from earlier runs. |
Resolution is dependent on the sample volume. Keep sample volume constant when repeating runs. |
General advice to achieve good performance
Before using the column make sure that:
- Correct system has been selected in UNICORN System Control
- Correct wavelength has been set for UV/UPC monitor
- All tubing has been properly connected and tubing is not longer than needed
- All connectors are free from leakage, verified by passing a leakage test
- No tubing is folded or twisted
- Online filter, if used, is changed on a regular basis
- Correct buffers are used for the chosen columns and proteins
- All inlet tubing has been immersed in correct buffer solutions
- Enough buffer has been prepared
- Buffers have been equilibrated to the environment temperature
- Buffers/eluents have been degassed if necessary (e.g., in RPC runs)
- Suitable columns have been selected for the target proteins
- Column meets pressure requirements for selected medium
- Columns have been cleaned and prepared according to column instructions
- Samples have been clarified by centrifugation and/or filtration prior to sample loading
- Samples have been adjusted to binding buffer conditions
- Auto sampler (if used) has been prepared according to user manual
- The fraction collector has been filled with appropriate number of microtiter plates or tubes
- Appropriate arrangement for waste handling has been prepared
Unusual column appearance
| 可能原因 | 建议的补救措施。 |
|---|---|
Unwanted air bubble formation. |
A restrictor in the chromatography system can prevent this. If a flow restrictor is attached, it is important to change the pressure limit to 0.5 MPa (5 bar) on the ÄKTAdesign system (where the column and the flow restrictor give a pressure of 0.3 MPa and 0.2 MPa respectively). |
Air trapped in the column. |
Allow thermal equilibration before use. |


