OptiRun™ Service solutions: Instrument qualification
Establish and maintain a qualified state
The importance of maintaining a qualified state
Maintaining compliance with regulatory bodies is a critical endeavor for all involved in the life sciences community. As a life sciences partner, we are dedicated to supporting compliance through qualification services. With updated, flexible and compliant qualification solutions for our customers, our global qualification services team understands how regulations affect the installation and utilization of the equipment we design and manufacture.
Learn more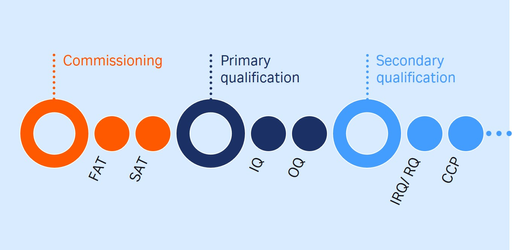
Qualified research starts with qualified expertise
Qualification Services take the guesswork out of what needs to be done for instrument qualification and requalification. We provide you with flexible solutions and the latest in regulatory knowledge. The suite of qualification services combines the latest product updates, service expertise, and technology available only from the original equipment manufacturer. Discover how we can help simplify the qualification of your instruments.
Contact us
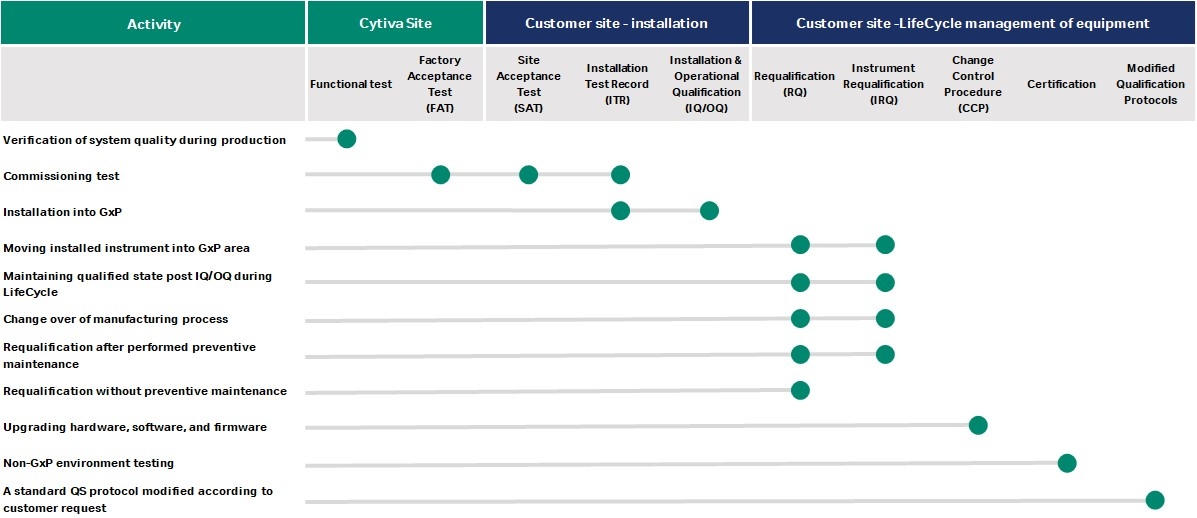
Why does requalification matter?
Changes can bring your instruments to an unqualified state — creating risk for your operation, with the potential to:
- lose production batches.
- waste time and money.
- fail to meet production or research deadlines.
To properly maintain the qualified state of your instruments, regularly perform preventive maintenance and requalification. Our team will also do periodic updates to hardware, software, or firmware, and you’ll have access to change control notifications as documentation.
Contact us
Recommended qualification services products
Simplify the qualification of your equipment with the service that’s right for you – find it in the table.
Contact us