-
Spotlight on vaccine production
Explore how working with qualified suppliers can speed innovation.
-
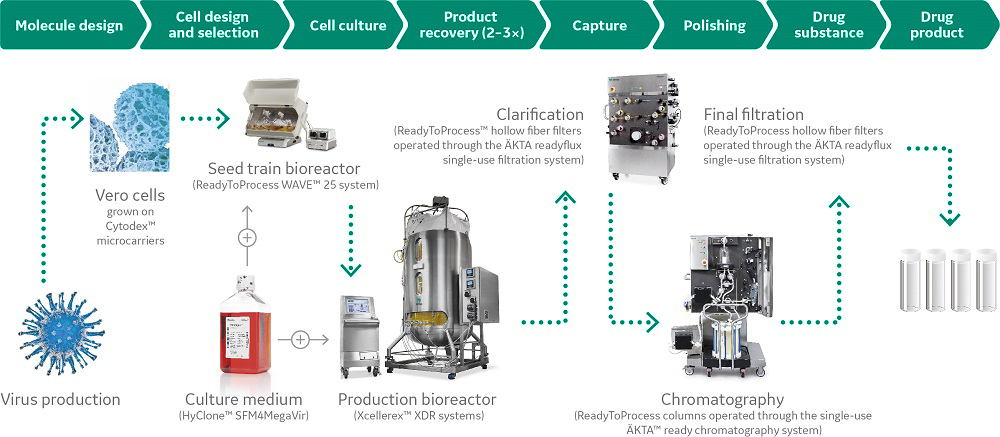
Platform solutions
Applying Quality by Design methodology to platform development is important for process success.
-
A balancing act
How can businesses ensure profitability while maintaining low prices for developing countries?
-
Let’s talk
We’re happy to discuss ways to overcome your vaccine development or manufacturing challenges.
- Application note: Scalable process for adenovirus production. Cytiva, KA877080618AN (2018).
- Application note: Process economic simulation for scalable production of adenovirus, Cytiva, KA3941080618AN (2018).
- Application note: Accelerated manufacturing of subunit vaccines for a rapid pandemic response. Cytiva, KA2393220218AN (2018)
- Application note: Process economy of using Cytodex Gamma microcarriers. Cytiva, KA1887150218AN (2018)
- Application note: Efficient purification of the pertussis antigens toxin, filamentous haemagglutinin, and pertactin in chromatography workflows. Cytiva, 29227789, Edition AA (2016)
- Whitepaper: Accelerate flavivirus vaccine production with modern tools and solutions (29236595AA)
- Whitepaper: Platform approach to purification of bacterial capsular polysaccharides for vaccine production (KA2040220118WP)
- Whitepaper: Overview of a scale-up of a cell-based influenza virus production process using single-use bioprocessing equipment (29043551AC)